Plastic Bottle Manufacturers in UAE – Poly Cos
LEADING PLASTIC MANUFACTURER IN UAE
- Sun - Fri: 9AM - 6PM
- India | UAE | Oman | China
- 0526029353
- India | UAE | Oman

Why Plastic Containers Affect Drug Shelf Life in Plastic Packaging
Why Plastic Containers Affect Drug Shelf Life in Plastic Packaging

Your antibiotic loses potency. Your cough syrup darkens. Your vitamin supplement degrades faster than expected.
The culprit? The plastic bottle.
Not all plastic containers protect drugs equally. Some fail catastrophically causing 10-20% potency loss per year. Others succeed. The difference comes down to material science, manufacturing precision, and climate reality.
Plastic packaging plays a critical role in maintaining drug shelf life plastic stability, especially in hot climates like the UAE and GCC region. High temperature, oxygen exposure, and moisture penetration can gradually reduce the effectiveness of pharmaceutical products during storage and transport.
Here’s what every pharmaceutical manufacturer needs to know.
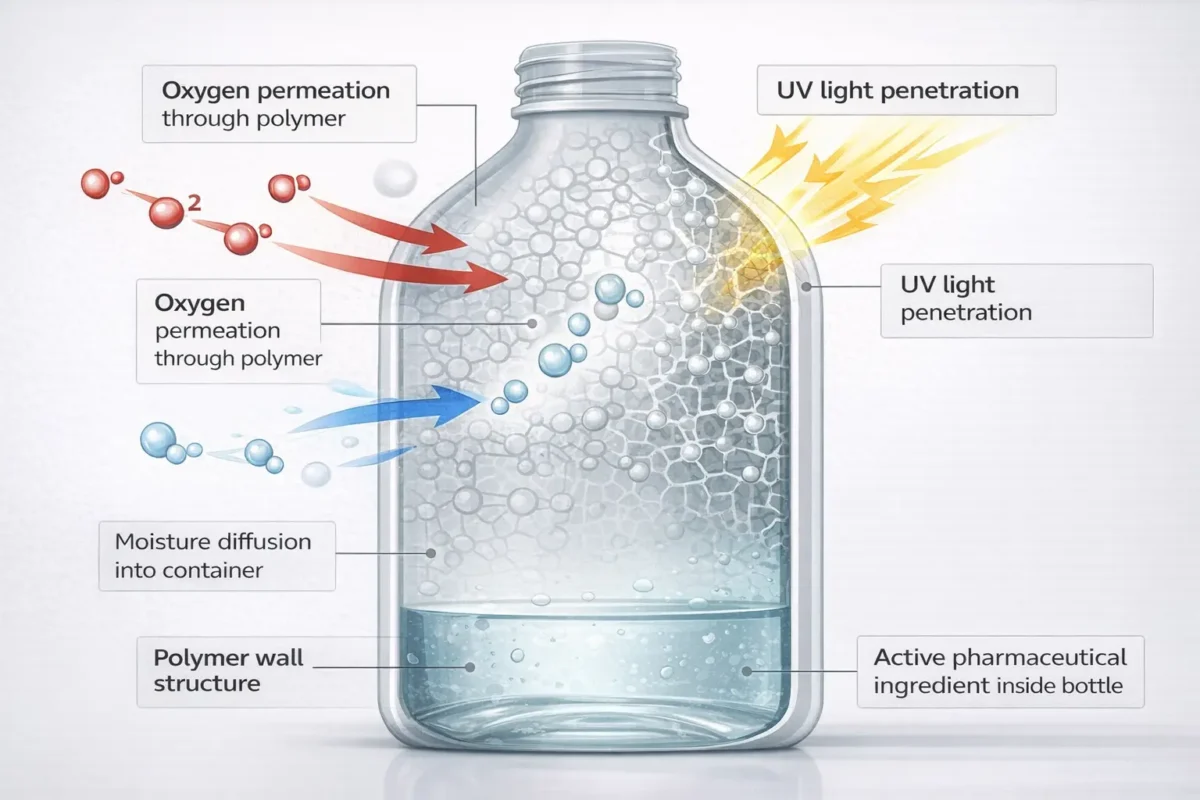
The Problem: Plastic Isn’t a Perfect Barrier
When you package a liquid medicine in plastic, three things happen immediately:
Oxygen Enters the Bottle
- Oxygen penetrates through plastic walls
- Reacts with your active ingredients
- Breaks chemical bonds
- Makes antibiotics less effective
- Causes vitamin C oxidation
- Result: Shortened shelf life
Moisture Moves Through Polymers
- Water vapor diffuses through container material
- Hygroscopic drugs (water-attracting) lose potency
- Tablets swell and lose effectiveness
- Powders cake and become unusable
- Result: Batch failure
Light Passes Through Transparent Bottles
- UV radiation above 320 nanometers damages drugs
- Photosensitive drugs degrade visibly
- Eye drops, antibiotics, vitamins turn yellow/brown
- Active ingredients break down
- Result: Loss of therapeutic benefit
The industry standard? Drug potency must remain above 90% at the expiration date. Anything less is a failed batch.
Why PET and HDPE Perform Differently
Material selection matters a lot.
Different drugs have different enemies. Some fear oxygen. Others fear moisture. Your container must protect against what your drug needs protection from.
PET Bottles: The Oxygen Blocker
What PET does well:
- Oxygen transmission rate: 1-5 cc/m²/day (excellent barrier)
- Keeps oxygen OUT effectively
- Transparent (good for visual inspection)
- Cost-effective for high-volume manufacturing
- Strong and durable
Best for:
- Antibiotics (oxidation-sensitive)
- Cough syrups (require oxygen protection)
- Vitamin solutions (prone to oxidation)
- Any drug where oxygen = enemy
PET’s weakness:
- Moisture permeability is higher
- Water vapor passes through more easily
- Not ideal for hygroscopic drugs (ones that attract water)
- Can cause potency loss in moisture-sensitive formulations
Real impact: If your drug is sensitive to moisture, a PET bottle fails. Your shelf life drops. Your customers notice. Your reputation suffers.
HDPE Bottles: The Moisture Blocker
What HDPE does well:
- Moisture vapor transmission: LOW (excellent barrier)
- Keeps water vapor OUT effectively
- Chemical-resistant (handles harsh formulations)
- Opaque (protects light-sensitive drugs)
- Impact-resistant
Best for:
- Syrups and suspensions (hygroscopic formulations)
- Liquid medicines that absorb moisture
- Products requiring chemical resistance
- Drugs sensitive to water ingress
HDPE’s weakness:
- Oxygen transmission rate: 100-200 cc/m²/day (poor barrier)
- Oxygen passes through more easily
- Not suitable for oxygen-sensitive drugs
- Can cause oxidation and rapid degradation
Real impact: If your drug is sensitive to oxygen, an HDPE bottle fails. Your active ingredient oxidizes. Potency drops by 10-20% per year. Your shelf life shrinks dramatically.
The Comparison
| Property | PET | HDPE | Winner |
| Oxygen barrier | 1-5 cc/m²/day | 100-200 cc/m²/day | PET (100x better) |
| Moisture barrier | Medium | High | HDPE (3-5x better) |
| Transparency | Clear | Opaque | PET (visual inspection) |
| Cost per unit | Low-Medium | Low | HDPE (slightly cheaper) |
| Weight | Light | Light | Tie |
| Best use case | Antibiotics, Syrups | Suspensions, Hygroscopic | Depends on drug |

Glass vs. Plastic (Why You Probably Can’t Use Glass)
Glass wins on every technical metric.
Glass advantages:
- No permeability (inert, impermeable)
- No extractables/ leachables risk
- Longest shelf life (20+ years)
- Ultimate drug protection
But here’s why pharma manufacturers use plastic instead:
| Factor | Glass | Plastic |
| Cost per unit | 3-5x more expensive | Standard cost |
| Weight (shipping) | Heavy (expensive logistics) | Light (cheap shipping) |
| Breakage risk | High (breakage = waste) | Zero (unbreakable) |
| Regulatory approval | Complex | Faster approval |
| Hot-climate transport | Okay | Ideal for 45°C+ |
| Manufacturing scalability | Limited | Unlimited |
The reality: Glass is perfect in a climate-controlled warehouse. In a 45°C shipping container heading to Saudi Arabia or Nigeria? Plastic is the only practical choice.
PET and HDPE aren’t better or worse.
They’re different.
Your job: Match the container to the drug’s enemy.
Get that right, and your shelf life stays intact.
Chemical Migration: The Hidden Risk
Here’s what most pharmaceutical manufacturers discover too late.
Your plastic container isn’t chemically inert.
What’s Inside Your Plastic Bottle
Inside the bottle, plastic releases small molecules called extractables. These include:
- Plasticizers — flexibility agents
- Stabilizers — prevent degradation
- Colorants — dyes and pigments
- Antioxidants — prevent oxidation
- Processing residues — from manufacturing
What Happens Next
When these extractables migrate into your drug formulation, they become leachables.
- They react with active ingredients
- They reduce drug potency
- They introduce safety risks
- They accelerate degradation
This happens FASTER at elevated temperatures.
A container stable at 25°C might leak significant amounts at 40°C—the standard accelerated stability test condition used by regulators worldwide.
The Regulatory Reality
USP standards <1663> and <1664> require manufacturers to test for extractables and leachables.
Skip this step, and your product fails regulatory approval.
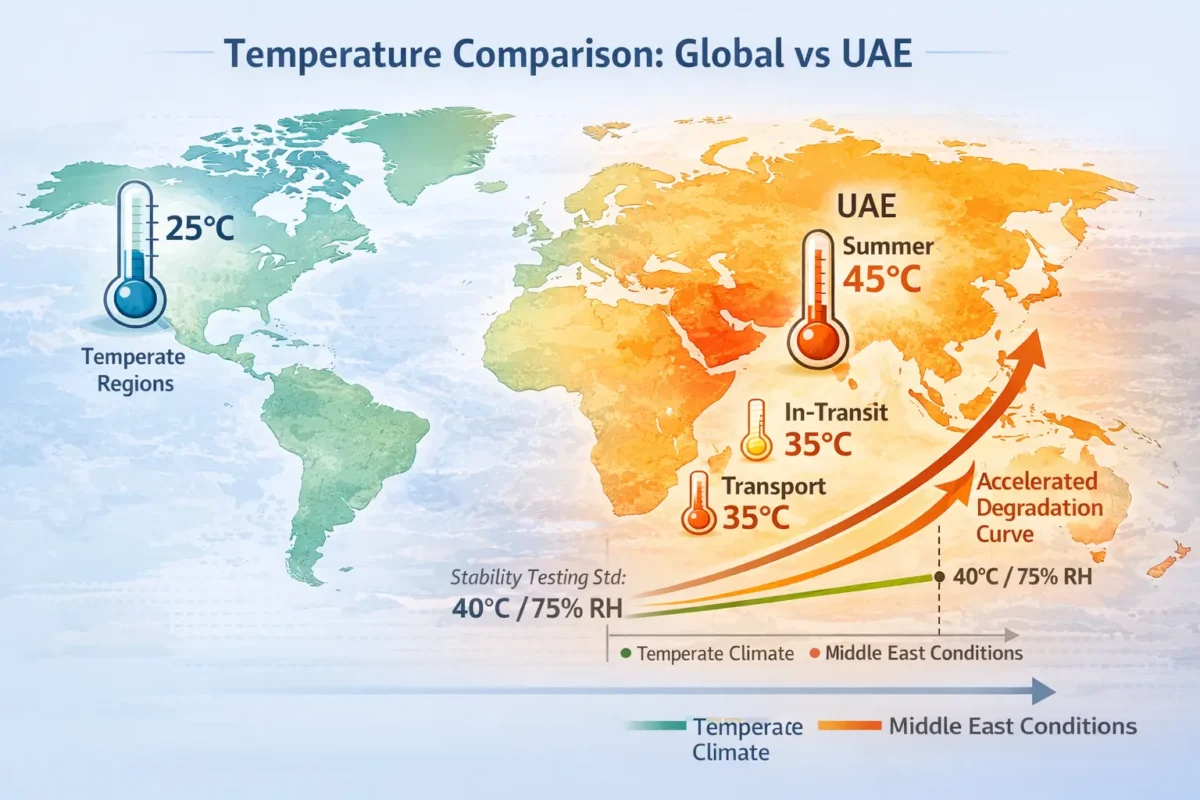
Temperature and Climate: The Middle East Challenge
This is where most guides fall short.
If you manufacture in the UAE, Saudi Arabia, or Africa, you face conditions most temperate-zone manufacturers ignore.
Temperature fluctuations directly impact drug shelf life plastic containers are designed to protect.
Real-World Conditions (Not Theory)
Summer warehouse temperatures:
- 35-40°C sustained (not climate-controlled)
- Interior containers reach even higher
- Equipment generates additional heat
In-transit conditions:
- 45-50°C inside shipping containers
- Some routes exceed 55°C in extreme climates
- Extended transit times amplify exposure
Stability testing standard:
- 40°C and 75% relative humidity (ICH guideline)
- Mimics real Middle East conditions
- Validates long-term performance
What Temperature Does to Permeability
At elevated temperatures, permeability rates accelerate exponentially.
- A container designed for 25°C storage fails in the Middle East
- Wall thickness becomes critical
- Material selection becomes non-negotiable
- Standard designs don’t survive real conditions
The Pharmaceutical Client Reality
Our pharmaceutical clients in the region require bottles engineered specifically for hot-climate distribution.
Standard designs don’t cut it.
Manufacturing Controls That Protect Shelf Life
Here’s what separates quality manufacturers from suppliers cutting corners.
Control 1: Wall Thickness Uniformity
Precision requirement: ±0.1mm
- Thin spots = faster permeability = shelf-life failure
- Thick spots = waste = higher costs
- Precision blow molding maintains consistency
- Every bottle meets the same standard
Impact: Consistent shelf life across your entire batch.
Control 2: Cleanroom Production
Contamination accelerates degradation.
- Pharmaceutical-grade manufacturing happens in controlled environments
- Continuous air filtration removes particles
- Zero-tolerance for foreign materials
- Every bottle meets pharmaceutical standards
Impact: No unexpected contamination causes batch failures.
Control 3: Pharmaceutical-Grade Resin Selection
Not all PET is equal.
- Pharmaceutical-grade resins are pre-tested for extractables
- Tested again post-production
- Food-grade or bottle-grade plastic doesn’t meet pharmaceutical standards
- Only approved resins enter production
Impact: Extractables stay below regulatory limits.
Control 4: Batch Traceability Systems
If stability fails, you need answers fast.
Traceability allows you to identify:
- Which production batch made the bottle
- Which resin lot was used
- Which facility produced it
- When it was manufactured
- How it was tested
Impact: Swift recalls and root-cause analysis. Minimal damage.
Control 5: Leak Testing & Pressure Testing
Every batch is validated.
- ✓ Microscopic cracks = shelf-life failure
- ✓ Modern testing detects failures at the 10⁻¹⁰ mbar level
- ✓ 100% inspection (not random sampling)
- ✓ Failed bottles never reach your warehouse
Impact: Zero surprises. Every bottle is verified.
The Cost of These Controls
These controls cost more. They take time.
But they’re the difference between:
- ✓ A product that lasts 3 years (with proper controls)
- ✗ A product that lasts 6 months (without controls)
That’s 6x the value.
How to Evaluate Your Pharmaceutical Bottle Supplier
Ask these six questions before committing:
- What’s your oxygen transmission rate at 40°C? (Target: <10 cc/m²/day for antibiotics)
- What’s your WVTR (moisture vapor transmission) at 75% RH? (Target: <0.5 g/m²/day for hygroscopic drugs)
- Do you conduct USP <1663> extractables testing? (Yes or no—there’s no middle ground)
- Do you have stability data in hot-climate conditions? (Not all suppliers do)
- How detailed is your manufacturing traceability system? (Batch-to-bottle linkage matters)
- What’s the scope of your GMP certification? (Which regions? Which facilities?)
Poor answers is a risk. Your shelf-life failures become your liability, not the manufacturer’s.
The Manufacturing Advantage: Why Poly Cos Delivers
The Manufacturing Advantage
At Poly Cos, we design bottles specifically for pharmaceutical stability. Our facilities in Sharjah, Ras Al Khaimah, Kuwait, and China follow pharmaceutical-grade protocols.
We know wall thickness matters. We test every batch. We document everything. And we understand hot-climate distribution—because we manufacture in the Middle East for Middle Eastern markets.
Our 10ml, 100ml, and 500ml pharmaceutical bottles are engineered for the conditions your drugs face. Not theoretical storage conditions. Real conditions.
The Bottom Line
Plastic containers reduce drug shelf life when:
- Material permeability exceeds drug tolerance limits
- Extractables and leachables migrate during storage
- Manufacturing quality controls are insufficient
- Design doesn’t account for regional climate conditions
But when you choose the right material, verify extractables/ leachables compatibility, and partner with a manufacturer who understands pharmaceutical-grade production, your drugs remain stable for their full shelf life.
Your shelf life isn’t determined by the expiration date you print. It’s determined by the container you choose.
Choose wisely.
Ready to Get Started?
Explore Your Options
Your pharmaceutical containers should be designed for stability. Not promises. Not theories. Real shelf-life protection in real-world conditions.
Selecting the correct material is essential to preserve drug shelf life plastic packaging systems must maintain under extreme transport conditions.
Poly Cos manufactures pharmaceutical bottles for manufacturers who can’t afford failures.
What we do:
- Test extractables and leachables (USP <1663>)
- Validate hot-climate performance
- Trace every bottle to its production batch
- Follow pharmaceutical-grade protocols
- Engineer for your specific climate region